Audio-book Review
By Chet Yarbrough
(Blog:awalkingdelight)
Website: chetyarbrough.blog
Great Courses Lecture Series
Chemistry and Our Universe
Presented by Ron B. Davis Jr., Ph.D. Associate Teaching Professor of Chemistry Georgetown University

Ron B. Davis Jr. ( Associate Teaching Professor of Chemistry Georgetown University)
Professor Davis’s lectures successfully interest a layman in the field of chemistry. However, this is not a simple introduction to chemistry.
Davis’s lectures are well done. Davis provides a lot of information with a summary of what has been said at the end of each lecture. His lectures offer interesting facts about chemistry, the world’s origin; its survival, and hopeful continuation. Where it loses some of its utility for a non-chemist is in calculations for chemical reaction, equilibration, and energy expenditure. Not that these calculations are not important, but they become too detailed for the merely curious.

At best, Davis’s lectures will spark a dilettante’s interest; at worst, they will lead a non-chemist to look elsewhere for easier understanding of the subject.
In his first 6 lectures, Davis breaks the science of chemistry into its most elementary particles.
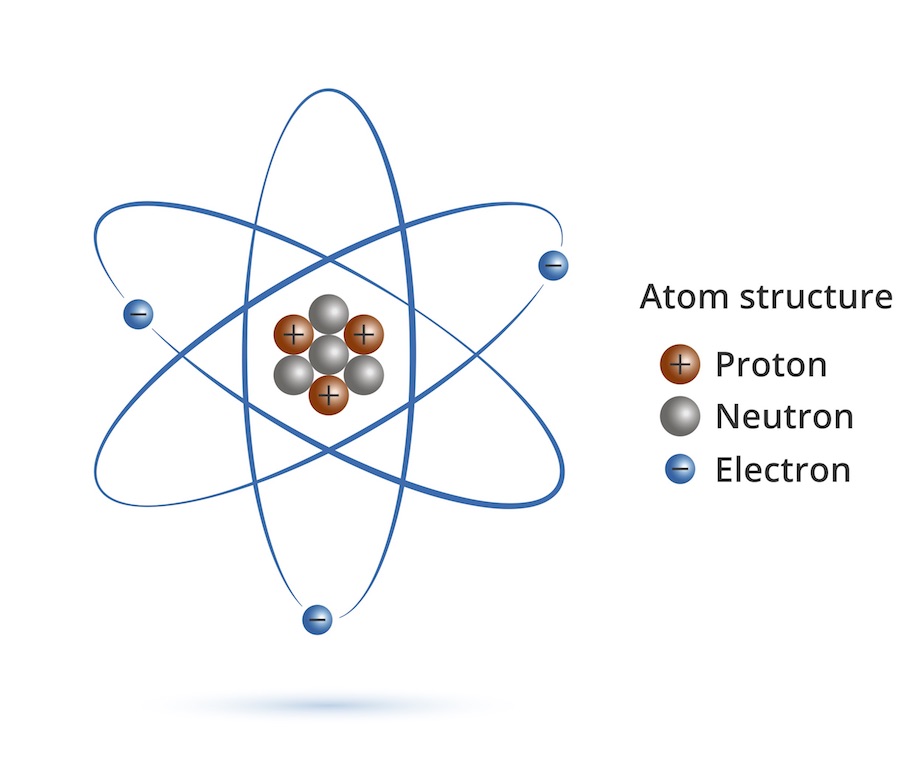
The role of atoms in chemistry is explained at the sub-atomic level of neutrons, protons, and energy producing electrons.
On the 7th lecture, Davis notes the fascinating history and utility of the periodic table. As of 2002, the known elements on the periodic table come to 118. Over 90 of those elements are naturally occurring. They are organized in the periodic table by number of protons (atomic number) in each nucleus with hydrogen being first (no. 1) and oganesson (a synthetic chemical) being the last (no. 118).
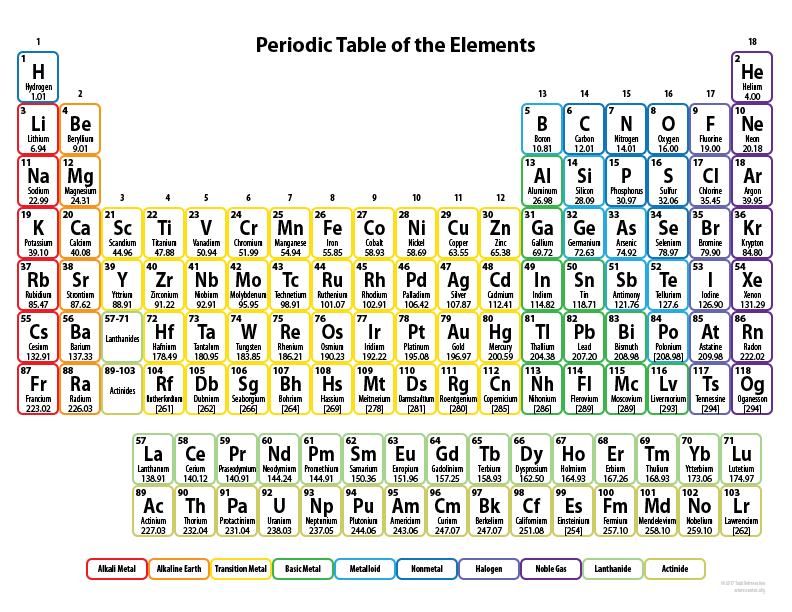
Davis explains the importance of the periodic table by their grouping. The groups are vertically and horizontally organized with each family of atoms beginning in a line from left to right and in columns from top to bottom.
The horizontal line reflects one of seven periods. Each atom in these periods has the same number of electron shells (aka orbitals) constituting the same energy level. To a degree, the horizontal rows share the same chemical characteristics.
The vertical rows begin with hydrogen and are generally classified as groups. The first group is classified as Alkali metals. The far right which is row 18 are called noble gasses; beginning with helium.
Elements in the same family (the vertical row) have the same electron configuration in their outer shell. A shell is what is called a valence shell, an orbiting electron around a nucleus. Elements in the same family tend to have a shared chemistry.
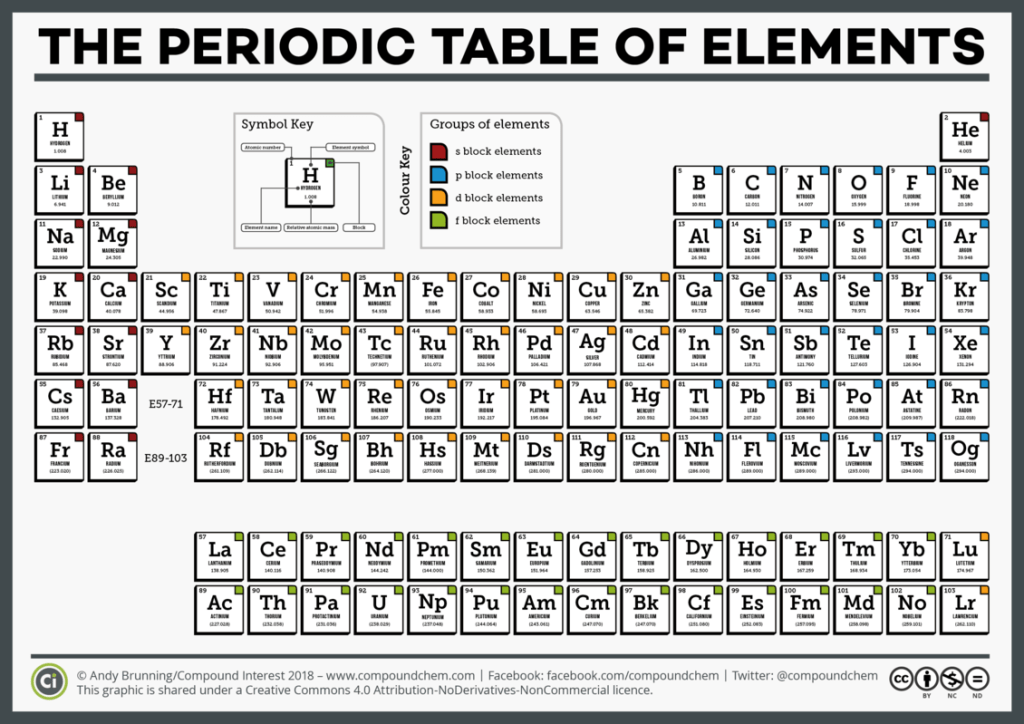
Davis notes that the vertical orientation of the periodic table indicates electrons get farther from the nucleus as you go down the table. The effect is to lower ionization energy as you go down the group; making electrons more easily released to other atoms. However, Davis notes this is not an iron clad rule; i.e. there are exceptions.

The next several lectures deal with chemical reactions, formations, and randomness and how they can be calculated. You enter the realm of chemistry mathematics. This is where listening for some of us meets ignorance, and understanding escapes.
Energy generation reawakens a listener’s interest. Davis explores the history of nuclear fission and fusion.
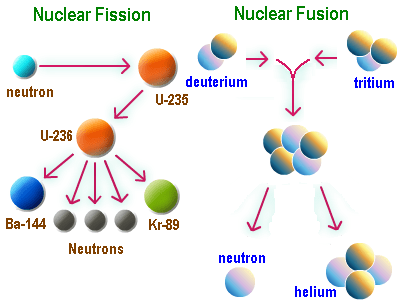

He explains the great promise and threat of Einstein’s insight to the equivalence of energy and mass. Fission leads to the destructive force of atomic bombs during WWII.
Nagasaki and Hiroshima show what uncontrolled fission can do in time of war.


Fukushima shows what uncontrolled fission can do in time of peace.
Three Mile Island, Chernobyl, and Fukushima were uncontrolled fission events that destroyed buildings and contaminated the environment. To date, fission has been found to be a viable source of energy but admittedly dangerous, and in disasters, highly polluting.
The principle of nuclear fusion may be the solution for a non-polluting, less dangerous, supply of energy.
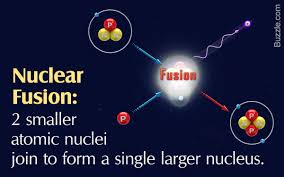
However, fusion has not been achieved because of the high energy demand (heat) required to compel atoms to fuse. Only the heat of the sun has successfully produced fusion. The hope is that a method of cold fusion will be discovered. The fundamental point made by Davis is that the production of nuclear energy is all chemistry.
In the next lectures, Davis addresses polymers, medicinal chemistry, poisons, chemical weapons, fuels, and explosives.

Davis explains why trees grow to be over a hundred feet tall while not being overturned by weather. It is largely due to cellulose which is one of the longest polymers in nature.
Davis notes DNA is one of the most complex of the natural polymers in chemistry. DNA contains all of the characteristics of carbon-based life forms.

He also notes chemistry is used to directly attack or fool human cancers that invade human DNA. These compounds have the potential for curing cancer.
Poisons are next. Davis notes that poisons have been around since the beginning of recorded history. He explains there are three classifications for this category of chemicals; e.g. poisons, toxins, and venoms.
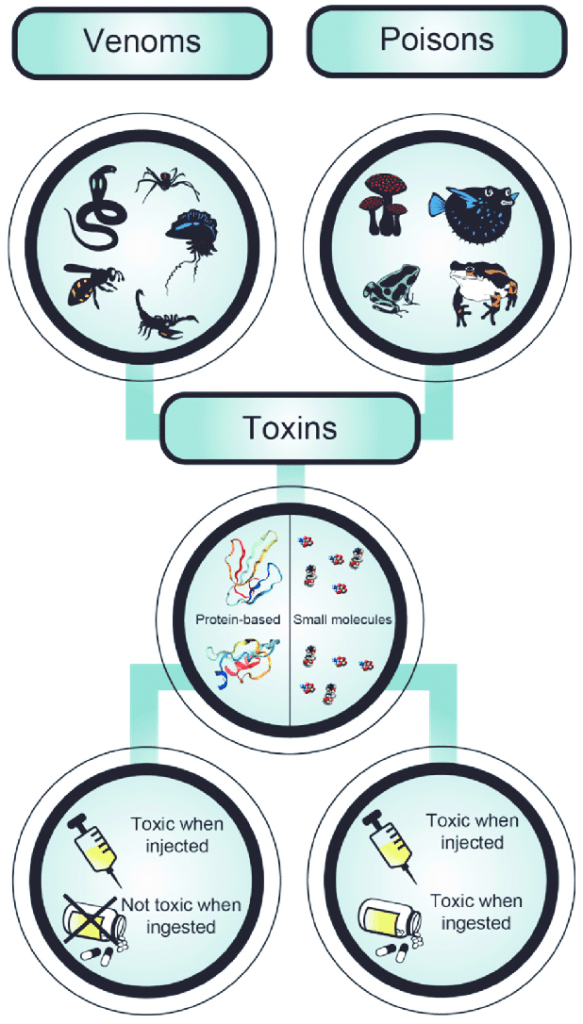
Poisons are substances that cause death, injury, or harm at a molecular level. The key to their effect is dosage. Toxins are substances produced within living cells that are contracted by touching or ingesting plants, or by contact with animals carrying microorganisms that cause disease. Davis explains venoms are secretions produced by animal’ or insect’ predators for defense or predation.
Fuels are lightly touched on by Davis with an examination of the discovery of fire; beginning with wood and progressing through other carbon-based materials.
Davis notes the evolution of coal, oil, and plant-based derivatives that produce fuel for industry and automobiles. He delves into the consequence of pollution in using these fuels, and their threat to humanity. He touches on global warming and its ecological consequence.

War is noted as impetus for the weaponization of chemicals, and explosives. A listener is introduced to Fritz Haber, a Jewish German genius that introduced chlorine gas (phosgene) to WWI. Haber became known as the father of chemical warfare.

Chemical warfare in Syria
Alfred Nobel, a Swedish chemist invented and patented dynamite. However, Davis notes high explosives were first discovered by Christian Schonbein in 1846. Schonbein’s discovery, like many chemical discoveries noted by Davis, is accidentally found when Schonbein spills hydrochloric acid in his lab and wipes it up with cloth apron. He puts the apron next to a stove to dry it out and it bursts into flame.
Alfred Nobel (Swedish chemist who invented dynamite and plastic explosives.)

Gunpowder cotton is discovered with Schonbein’s accident. The problem is that gunpowder cotton was too volatile. It is left to Nobel to come up with a stabilizing compound for gunpowder cotton to create sticks of dynamite. Nobel invents an igniter fuse to start the explosive potential of dynamite. Further discovery by Nobel leads to nitroglycerin and plastic explosives in the late 19th century.
Davis ends his lectures with the chemistry of earth. He notes how life may have begun with chemical building blocks introduced to earth from fragments of meteorites. Meteorites are created from exploding stars. Where water exists, he argues organic life is possible. Davis concludes–human exploration of the Universe holds hope for the future.

Davis speculates that there could very well be a habitable planet that has the same characteristics as early earth. He suggests, with the building blocks of life coming from meteorites, only water needs to be added to create and sustain life.

